參賽序號:27
海報主題
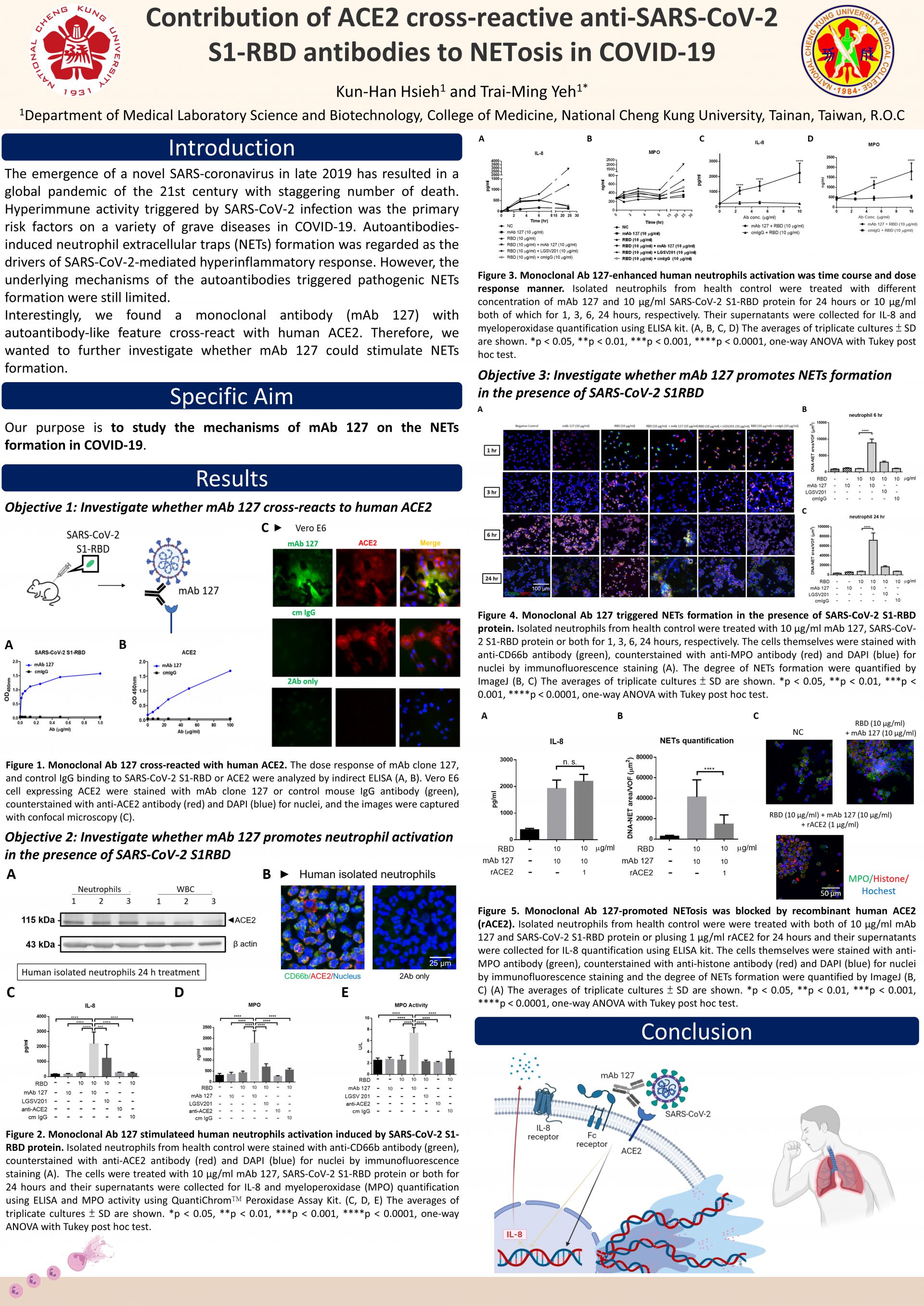
Contribution of ACE2 cross-reactive anti-SARS-CoV-2 S1-RBD antibodies to NETosis in COVID-19
系級
醫學檢驗生物技術學系
指導老師及參賽學生
指導老師:葉才明
參賽學生:謝坤翰
構想說明
COVID-19, which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, has posed a serious threat to global health. The disease not only causes acute respiratory diseases affecting the overall physiological function but also brought out many unpredictable complications. Hyperactivation of immune response and the production of autoantibodies triggered by SARS-CoV-2 infection were two primary risk factors on a variety of grave diseases in COVID-19. Among them, neutrophil extracellular traps (NETs) formation was regarded as one of the drivers of SARS-CoV-2-mediated hyperinflammatory response. Recent evidence exhibited that autoantibodies and immune complex, promoted the pathogenic NETs formation and further accelerated the disease progression in COVID-19 patients. However, the underlying mechanisms of the autoantibodies triggered pathogenic NETs formation were still limited. Previously, our team found a monoclonal antibody (mAb 127) which can recognize both SARS-CoV-2 spike protein receptor-binding domain (S1-RBD) and human angiotensin-converting enzyme 2 (ACE2), a well-known receptor of SARS-CoV-2. However, the pathogenic roles of mAb 127 are unclear. In this study, we investigated whether mAb 127 could trigger NETs formation. We treated human isolated human neutrophils with mAb 127 in different conditions. The results showed that mAb 127 or SARS-CoV-2 S1-RBD alone could not give rise to remarkable impact on neutrophils activation. However, the levels of IL-8 secretion, myeloperoxidase activity, and NETs formation were dramatically increased in the S1-RBD and mAb 127 co-treated group, but not S1-RBD with control mouse IgG or common immune complex groups. Furthermore, NETs formation induced by mAb 127 was inhibited in the presence of recombinant ACE2. Taken together, our results suggested that autoantibodies recognizing both SARS-CoV-2 S1-RBD and human ACE2 may contribute to NETosis in COVID-19.

 114年活動,現正報名中!
114年活動,現正報名中!